Where Science Meets Innovation
At Helix Life Sciences, our team of experts collaborates closely to deliver quality-driven solutions,empowering your life science journey from development to market

Trusted Experts,
Proven Excellence
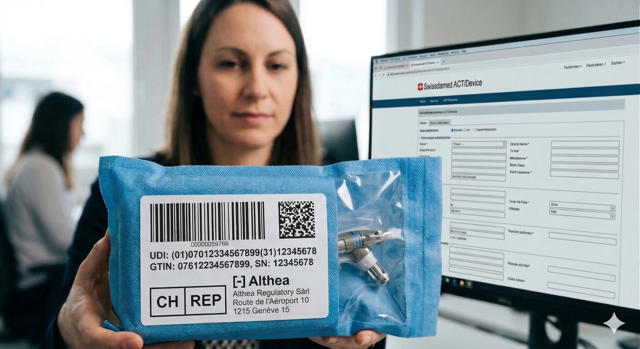
At Helix Life Sciences, trust is not just earned; it is woven into every strand of our expertise. Based in Geneva, at the heart of Switzerland’s international life science ecosystem, we support pharmaceutical, biotech, and medtech companies in navigating the complexities of Swiss regulatory frameworks.
Our team is composed of seasoned experts with deep scientific, regulatory, and quality management backgrounds, complemented by dedicated operational staff. Together, we offer tailor-made services that ensure fast, reliable, and compliant access to the Swiss market.
With years of hands-on experience across regulatory affairs, Swissmedic establishment licence applications, Qualified Person (QP/RP/FvP) services, and quality management systems (QMS), we bring clarity and confidence to every step of your market entry. Whether you are an international company establishing your Swiss presence or a local innovator scaling operations, our approach combines global standards in line with 2026 Swissmedic regulations.
Our Services
Why us
Years Of Experience & Industry Accreditations
1
Uphold high ethical standards and act with honesty, transparency, and professionalism in all your interactions.
2
Stay up-to-date with industry trends, regulations, and best practices to offer informed advice to your clients.
3
Our success is rooted in the collective expertise and dedication of our professional team.
Advancing Innovations For A Healthier Tomorrow

Articles
Valuable Insights To Empower Decision-making
Start Your Journey with Helix
Drop your contact details into the form, and we’ll reach out to you!