Explore Topics
- Featured / Analysis
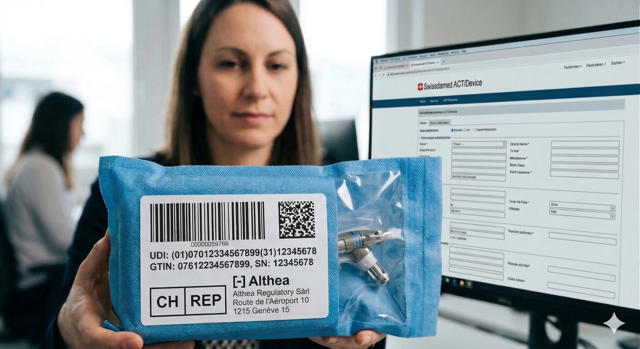
In the changing landscape of European MedTech, Switzerland plays a unique and complex role. With the termination of the Mutual Recognition Agreement (MRA) in May 2021, the Swiss market has transformed into a "third country,"...
- Featured / Analysis
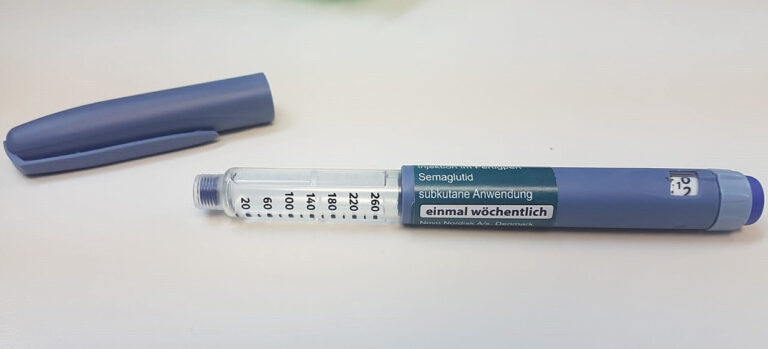
In a concerning development, counterfeit Ozempic pens have been discovered in the EU market, raising alarms about the safety of patients. These fake products, indistinguishable from genuine ones, have been traced back to wholesalers in...
- Featured / Analysis
As the pharmaceutical landscape continues to evolve, the need for standardized identification and management of medicinal products has never been more critical. The Identification of Medicinal Products (IDMP) standards, established by the International Organization for...
- Featured / Analysis
The European Medicines Verification System (EMVS) has been a game-changer in enhancing the safety and integrity of the pharmaceutical supply chain across Europe. By implementing a unique identifier (UI) for each medicinal product, EMVS facilitates...