Swissdamed
One of the most significant upcoming milestones is the full implementation of Swissdamed. Because Switzerland no longer has access to the central EU database (Eudamed), Swissmedic has developed its own platform to ensure market transparency and surveillance. Swissdamed is designed to mirror the functional modules of its European counterpart, beginning with the registration of economic operators and followed by the registration of devices.
The registration of actors – manufacturers, authorized representatives, and importers – is the first critical hurdle. The Actor Module (ACT) has been live since August 2024. All Swiss-based economic operators—manufacturers, authorized representatives, and importers—must register here to obtain their Swiss Single Registration Number (CHRN).
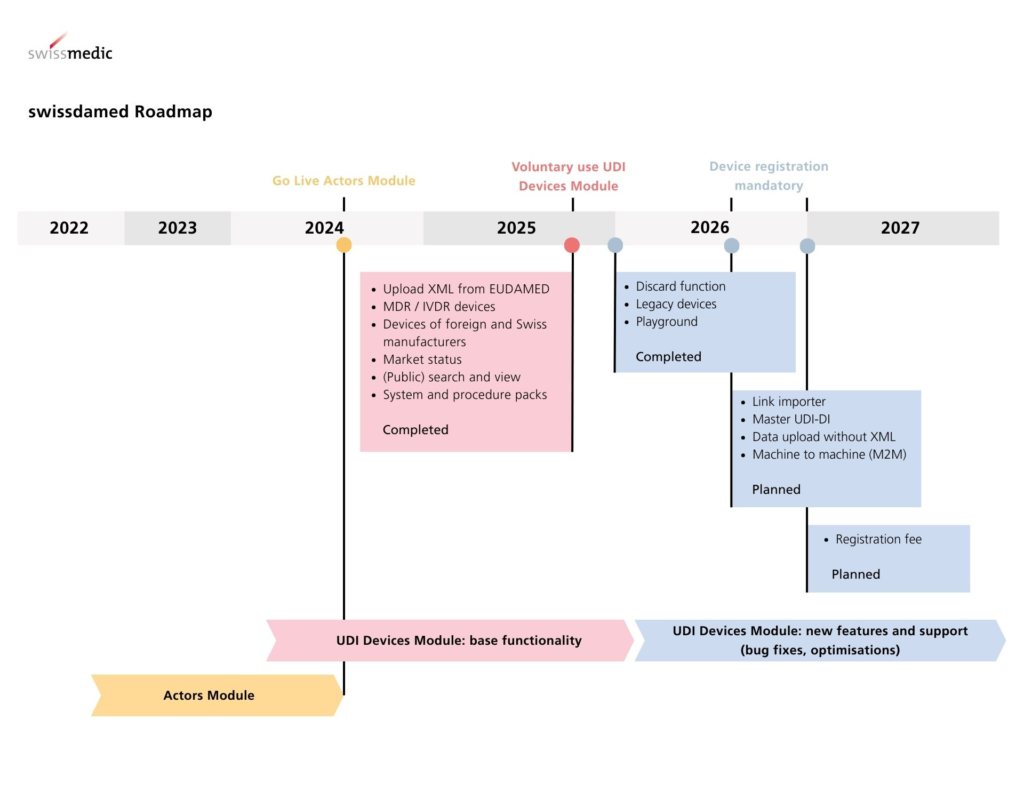
While some deadlines for economic operator registration have already passed, the rollout of the device registration module will necessitate a proactive data management strategy.
The Device Registration (UDI) Module became available for voluntary registration in August 2025. This allows companies to begin uploading data for medical devices, IVDs, and systems/procedure packs. While currently optional, July 1, 2026, marks the start of the mandatory registration phase. From this date, any new device placed on the Swiss market must be registered before its launch. For products already on the market, a transitional period exists until December 31, 2026, by which point all devices must be fully registered in Swissdamed.
Companies should not view Swissdamed as a mere clerical task; it is a legal prerequisite for market access. Ensuring that your data is accurate and synchronized with your technical documentation is essential to avoid delays in product placement or potential enforcement actions from Swissmedic.
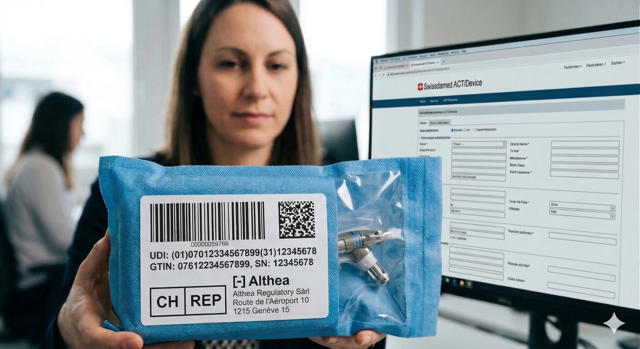
CH-REP
Central to this new Swiss framework is the role of the Swiss Authorised Representative (CH-REP). For any manufacturer based outside of Switzerland, the appointment of a CH-REP is not an optional convenience but a mandatory legal anchor. The CH-REP acts as the formal liaison between the manufacturer and the Swiss market surveillance authorities. Their responsibilities go beyond simply holding a certificate; they are legally liable for the device’s compliance. This includes verifying that the Declaration of Conformity and technical documentation have been drawn up, and ensuring that the manufacturer has fulfilled their registration obligations.
From a practical standpoint, the CH-REP must be integrated into the manufacturer’s quality management system. They must have permanent access to the technical documentation and be prepared to respond to inquiries from Swissmedic within short timeframes. For many companies, the transition periods for appointing a CH-REP have already lapsed for higher-risk classes, but for certain In Vitro Diagnostics (IVDs) and legacy devices, the window for compliance is narrowing. It is advisable to review current mandates to ensure the CH-REP is not just a name on a label, but a functional partner capable of managing the regulatory burden.
PRRC
Closely linked to the CH-REP is the requirement for a Person Responsible for Regulatory Compliance (PRRC). Under the Swiss Medical Devices Ordinance (MedDO) and the Ordinance on In Vitro Diagnostic Medical Devices (IvDO), both the manufacturer and the CH-REP must have a PRRC at their disposal. The PRRC’s mandate is clear: they must ensure that the conformity of the devices is appropriately checked, that the technical documentation and the EU declaration of conformity are kept up to date, and that post-market surveillance obligations are met.
The challenge for many organizations lies in the qualifications and the availability of the PRRC. This individual must possess the requisite professional knowledge, usually evidenced by a degree in a relevant scientific or legal discipline and several years of professional experience in regulatory affairs or quality management related to medical devices. In the Swiss context, the PRRC of the CH-REP ensures that the representative can fulfill their oversight duties effectively. This dual-layer of accountability—at the manufacturing level and the representative level—is designed to close gaps in market safety.
For companies operating in Switzerland, the immediate priority should be a comprehensive “gap analysis” of their Swiss-specific documentation. It is vital to ensure that labeling, Instructions for Use (IFU), and symbols correctly identify the CH-REP. Furthermore, manufacturers should begin preparing their data for Swissdamed registration now, rather than waiting for the platform’s final deadlines.
The Swiss market remains a lucrative and stable environment for medical innovation, but the cost of entry is now defined by administrative diligence. Compliance is not a static achievement but a continuous process of alignment with Swissmedic’s evolving digital and legal requirements. By securing a robust CH-REP partnership and clarifying the roles of the PRRC, manufacturers can mitigate the risks of market exclusion and focus on the primary goal: providing safe and effective technology to the Swiss market.