Fachtechnisch verantwortliche Person (FvP) & QP Mandates for the Swiss Market
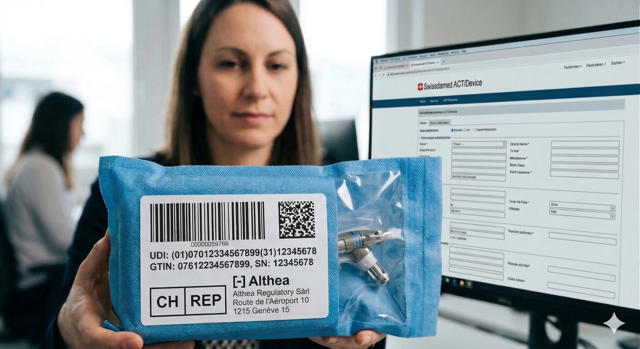
Navigating the Swiss therapeutic market requires a local legal anchor. Under the Swiss Arzneimittel-Bewilligungsverordnung (AMBV), every holder of an establishment license must appoint a Responsible person (QP). At Helix Life Sciences, we provide highly qualified FvP and Deputy (Stellvertreter) mandates that go beyond simple signature services to provide active, risk-based quality oversight and market release for your products.
Helix Life Sciences provides Responsible Person (Fachtechnisch verantwortliche Person – FvP) and deputy services in full compliance with Swiss law, as well as acting as the local Responsible Person for pharmacovigilance (QPPV). We also ensure seamless regulatory compliance by preparing, attending, and following up on Swissmedic inspections on behalf of our clients.

- Our Capabilities
Accelerate Your Business Development Plans
Active FvP & Deputy Mandates
Full legal representation for your Swiss subsidiary, ensuring 24/7/365 regulatory coverage.
Batch Release & QP Services
Final certification and market release for pharmaceutical products entering the Swiss healthcare system.
GDP & QMS Maintenance
Building and overseeing a Swiss-specific Quality Management System that meets local AMBV and AWV requirements.
- Swissmedic Compliance
Our QP/FvP Mandate Framework
(Compliance 2026)
In accordance with Swissmedic Technical Interpretation I-SMI.TI.17d, we ensure a minimum presence time, tailored to your company's risk profile
Our FvPs maintain a live list of critical steps (Audits, CAPAs, Validations) requiring onsite supervision.
We integrate the latest 2026 Swissmedic requirements for Signal Notification into your local QMS.

Expert Technical Oversight
Choosing the right legal anchor for your Swiss operations is a critical decision that directly impacts your long-term market stability. At Helix Life Sciences, we distinguish ourselves by offering an active, senior-led partnership that prioritizes operational integrity. Under the Swiss Arzneimittel-Bewilligungsverordnung (AMBV), every holder of an establishment license must appoint a Fachtechnisch verantwortliche Person (FvP). This role is a legal pillar that requires local residency and personal availability at the Swiss site to maintain effective quality oversight – a requirement that foreign residents cannot fulfill.
Located both in Zurich and at the heart of the Health Valley in Geneva, our team brings a deep understanding of the Swissmedic ecosystem and a proven success rate in facilitating inspections for foreign biotechs. We provide a rare hybrid expertise in both RP / FvP (Pharma) and PRRC (Medtech) mandates.
For companies beginning their Swiss market entry, the journey from initial QMS design to the successful completion of a Swissmedic onsite audit typically spans six to nine months. By partnering with Helix, you gain more than a signature; you gain a dedicated team committed to ensuring your Swiss subsidiary remains audit-ready and operationally resilient within the rigorous regulatory affairs landscape.
Start Your Journey with Helix
Drop your contact details into the form, and we’ll reach out to you!